Dear All,
I have created a box in lammps, with half of the box with Cu and other half with Ar. Initially I have created Cu fcc crystal pattern whereas Ar was also taken as fcc.And have assigned forcefield by—
units metal
group Ar type 2
group Cu type 1
mass 1 63.546
mass 2 39.948
pair_style hybrid eam/fs lj/cut 2.5
pair_coeff * * eam/fs Mendelev_Cu2_2012.eam.fs Cu NULL
pair_coeff 1 2 lj/cut 0.0650071 2.872 2.5
pair_coeff 2 2 lj/cut 0.0104233 3.405 2.5
Now, to eqilibriate the sample at ambient condition I employed fix NPT, after creating velocities for temperature 300K and pressure of 1atm,
timestep 0.001
velocity all create 300 12345 mom yes rot no
fix 1 all npt temp 300 300 0.1 iso 0 0 1
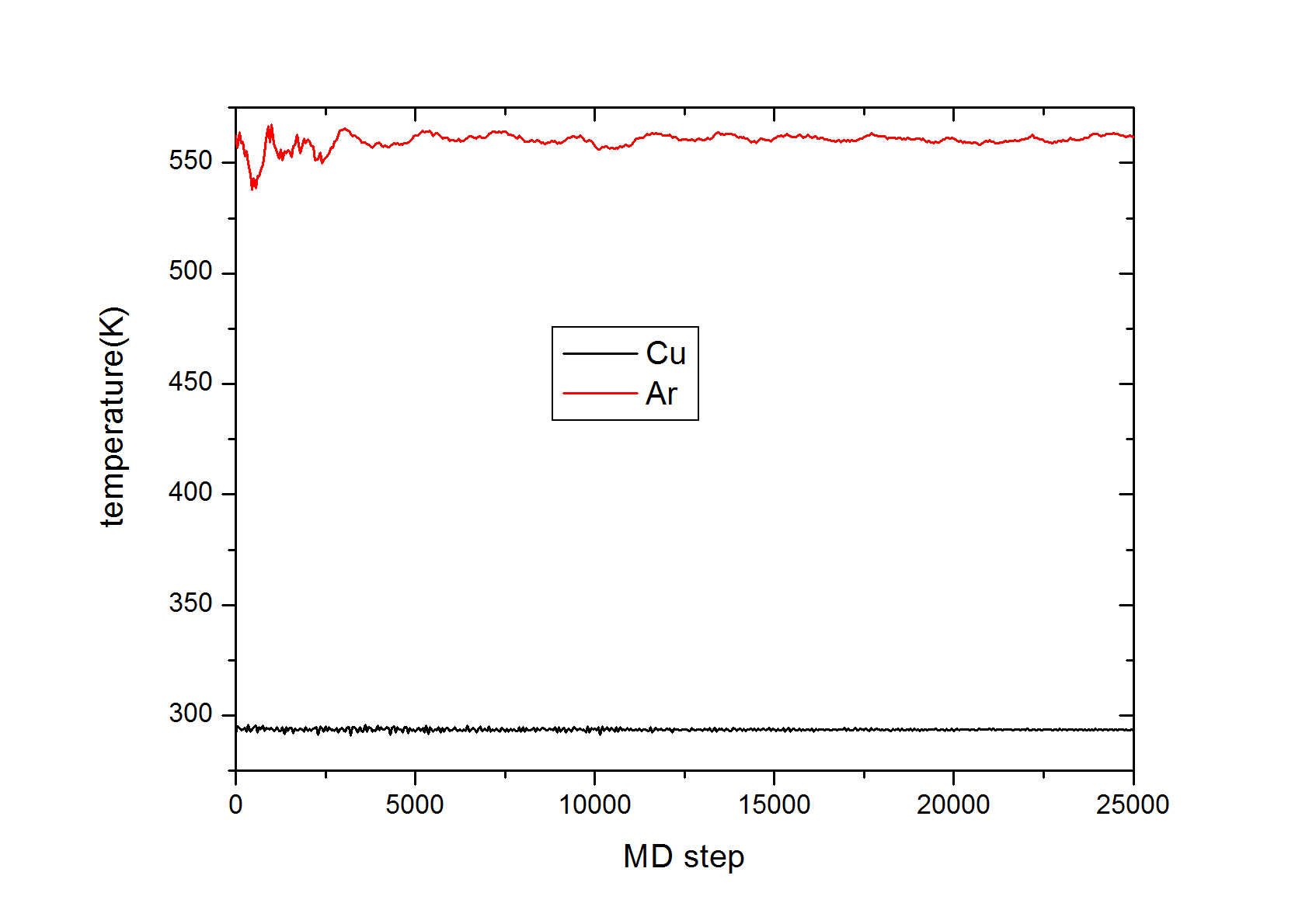
To get the temperature of Cu and Ar group separately, I used tem/partial with 1 1 1 for group Cu and Ar individually, but I get Ar temperature ~600K whereas Cu temperature is fine as per settings, it’s 300 K. Here is the plot for the last 2.5 ps.

Please make your comments/suggestions. Thanks in advance for your time.
What are your commands for the temperatures
of the 2 atom types and how you output
them to the log file (or however you are plotting them?)
Steve
to calculate the temperature of different group I wrote –
group Ar type 2
group Cu type 1
compute CuTemp Cu temp/partial 1 1 1
compute ArTemp Ar temp/partial 1 1 1
thermo_style custom step c_CuTemp c_ArTemp temp
then I just plot step vs c_Cutemp and c_ArTemp. But average CuTemp was ~300K where ArTemp was ~560 K. Also the overall temperature of the sample temp shows that it is 300 K. Is there any wrong to choose temp/partial for calculating temperature of two different groups ?
If you simply did this:
group Ar type 2
group Cu type 1
compute CuTemp Cu temp/partial 1 1 1
compute ArTemp Ar temp/partial 1 1 1
thermo_style custom step c_CuTemp c_ArTemp temp
and this:
velocity all create 300 12345 mom yes rot no
I don’t see how the time=0 temperature of Ar and Cu could
be different by 2x. Can you post a simple input script
that does nothing but setup your problem and a run 0
to print the initial temps and show that they are not 300?
Steve
I think I have explained the problem probably in wrong way.
I have checked that at timestep 0, Ar and Cu temperature prints ~300 K. But I applied
timestep 0.001 # in real unit
fix 1 all npt temp 300 300 0.01 iso 0 0 0.1
then after almost 2.5 ps Ar temperature goes up to 600K but Cu temp was 300 K.
The plot attached in the previous mail shows the second equilibration step i.e. from 2.5 ps to 5 ps. In the plot 2.5 ps is 0 in the plot.
I checked that at timestep 0, Cu temp Ar temp and temp prints ~300 K.
What may be the problem, please make your suggestion.
I think I have explained the problem probably in wrong way.
I have checked that at timestep 0, Ar and Cu temperature prints ~300 K.
But I applied
timestep 0.001 # in real unit
fix 1 all npt temp 300 300 0.01 iso 0 0 0.1
then after almost 2.5 ps Ar temperature goes up to 600K but Cu temp was
300 K.
The plot attached in the previous mail shows the second equilibration step
i.e. from 2.5 ps to 5 ps. In the plot 2.5 ps is 0 in the plot.
I checked that at timestep 0, Cu temp Ar temp and *temp* prints ~300 K.
What may be the problem, please make your suggestion.
a few picoseconds is nothingness in this context. equilibration times can
be much larger, especially when you need to transfer energy across phase
boundaries and have no heat dissipation through transport of particles.
if your initial configuration was not equally close to (or away from)
equilibrium for both species, the behavior you observe is to be expected.
please remember that you apply a thermostat to your *global* temperature,
not each subsystem.
now, how to do equilibration properly is less than a LAMMPS issue, but a
generic MD problem and like it was said many times here before, these are
questions that should be primarily discussed with your adviser and whoever
is training you in doing MD. it takes a (near) genius to pick this all up
by yourself and avoid all the pitfalls and mistakes and re-discover all the
solutions that people have discovered in over half a century of doing MD.
that being said, you should check via running in NVE ensemble where the
heat is coming from and avoid that and probably also use a dissipating
thermostat (temp/csvr or langevin) for the initial part of equilibration to
encourage the desired equipartitioning of kinetic energy.
axel.