I was wondering how do you determine the chemical potential of Li. The chemical potential of Li is the control variable in determining the grand potential. However, the chemical potential is dependent on the mole fraction of Li in a system: mu_Li= mu_Li_o+RT ln (activity) where activity= gamma(deviation from ideality)*x_Li
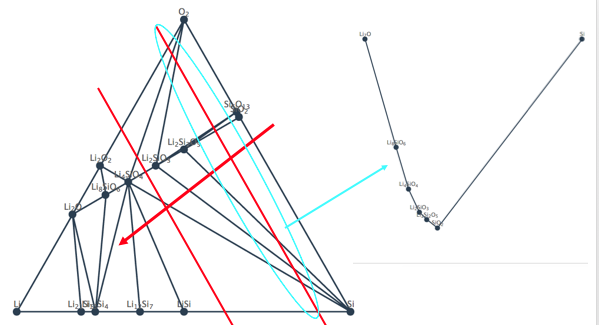
The way I understand the extraction of the stable phase at a given chemical potential from ternary phase diagram is like what I shown in the image attached. I move the red line towards the Li (increasing x_Li) and what the line cross determines the stable phases at that chemical potential of Li (aka x_Li). But it is not clear from what can of solution model you used to convert the x_Li to mu_Li.
Thank you in advance!